Introduction: A Process Too Often Misunderstood
You have aluminium parts that need to last. They need to resist corrosion, handle wear, and look professional but somewhere between the drawing board and the production floor, the finishing process becomes a black box. For many buyers, engineers, and business owners, the anodising process is one of the most misunderstood steps in aluminium surface treatment.
Too many procurement decisions are made without a clear picture of what anodising actually does, what types exist, and critically where quality can break down. This guide changes that. Whether you are new to the industry or simply want to sharpen your knowledge, this is your no-jargon, practical introduction to anodising.

Aluminium profiles undergoing the anodising process in an industrial surface treatment facility
What Is Anodising?
Anodising is an electrochemical process that converts the surface of aluminium into a durable, corrosion-resistant oxide layer. Unlike painting or plating where a coating is applied on top of the metal anodising grows the protective layer directly out of the aluminium itself. The result is a finish that cannot peel, chip, or flake.
The term "electrochemical" simply means the process uses electrical current and a chemical solution (usually sulphuric acid) to drive the transformation. The aluminium part acts as the positive electrode (the "anode"- which is where the name "anodising" comes from) in this controlled chemical reaction.
How Does the Anodising Process Work? Step-by-Step
The anodising process follows a precise sequence of stages. Each step matters- a failure at any point can compromise the entire batch.
Step 1 — Pre-Treatment (Cleaning)
Parts are cleaned to remove oils, dust, and surface contaminants. This typically involves alkaline degreasing, an acid etch (to create a uniform matte surface), and a desmut rinse (to remove metallic residues left by the etch). Clean metal is essential; any contamination will cause defects in the final finish.
Step 2 — Anodising (Oxide Layer Formation)
The cleaned parts are submerged in an electrolyte bath usually a dilute sulphuric acid solution and a direct electrical current is applied. The current causes oxygen ions from the electrolyte to combine with the aluminium surface, forming aluminium oxide (Al₂O₃). This oxide layer is porous at the microscopic level, which is important for the next stage.
Step 3 — Colouring (Optional)
If a colour is required, dyes are introduced into the porous oxide layer at this stage. The dye molecules become trapped within the microscopic pores, producing rich, stable colours that are far more durable than surface paints.
Step 4 — Sealing
The final stage closes the pores in the oxide layer, typically using hot deionised water or a nickel acetate solution. Sealing locks in any dye and significantly boosts corrosion resistance. A poorly sealed part will fail corrosion tests and look dull much sooner in service.
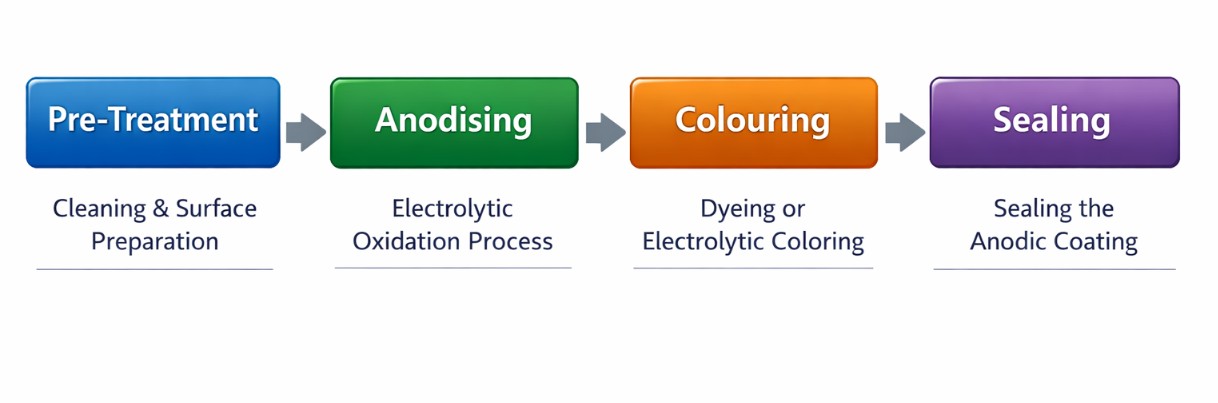
Step-by-step flowchart of the aluminium anodising process from pre-treatment to sealing
Types of Anodising: Which One Do You Need?
Not all anodising is the same. The right type of anodising depends on your performance requirements, environment, and budget. Here are the three main classifications defined by the MIL-A-8625 military specification the most widely referenced standard in the industry.
Type I — Chromic Acid Anodising
Uses chromic acid as the electrolyte. Produces the thinnest oxide layer (0.5–2.5 microns) and is the least common type due to environmental regulations around chromic acid. It is still used in aerospace applications where tight dimensional tolerances are required and where minimal material build-up is critical.
Type II — Sulphuric Acid Anodising
The industry workhorse. Uses sulphuric acid as the electrolyte and produces an oxide layer of 5–25 microns. It is the most widely used form of aluminium surface treatment across architecture, consumer electronics, automotive components, and general industrial parts. It offers a good balance of corrosion resistance, dye absorption, and cost.
Type III — Hardcoat Anodising
Also called "hard anodising." Runs at lower temperatures and higher current densities to produce a very thick, dense oxide layer of 25–150 microns. It delivers exceptional hardness (approaching that of industrial chrome), excellent wear resistance, and superior corrosion protection. Typical applications include hydraulic components, firearms, cookware, and military equipment.

Oxide layer thickness comparison across Type I, Type II, and Type III anodising — from thin chromic acid coatings to hard anodising up to 150 microns
Key Benefits of the Anodising Process
Anodising is a preferred aluminium surface treatment for good reason. Its advantages are practical and measurable:
- Corrosion resistance: The oxide layer acts as a barrier against moisture, salt, and atmospheric attack — critical for outdoor and coastal environments.
- Wear resistance: Especially in Type III, the hardened surface dramatically extends part life under mechanical friction.
- Aesthetics: Anodising produces consistent, attractive finishes across large batches with colour options that do not fade like paints.
- Dimensional stability: The layer grows into the surface, so part dimensions change minimally important for precision engineering.
- Electrical insulation: Aluminium oxide is non-conductive, making anodised parts suitable for electrical isolation applications.
- Recyclability: Anodised aluminium can be fully recycled without any special pre-treatment to remove the coating.

Anodised aluminium profiles in silver and bronze finish — demonstrating the aesthetic versatility and durability of the anodising process
Common Quality Challenges in Anodising Quality Control
Despite being a well-established process, anodising is sensitive to variation. Small deviations in bath chemistry, temperature, or racking technique can result in costly defects. Understanding these challenges is essential for anyone responsible for anodising quality control.
Bath Chemistry Drift
The concentration of sulphuric acid and dissolved aluminium in the bath changes continuously during production. If not monitored and corrected in real time, the oxide layer thickness and quality will vary between batches and sometimes even within a single batch.
Temperature Instability
Anodising baths must be held within a narrow temperature range (typically 18–22°C for Type II). Temperatures even a few degrees too high produce a "burned" or soft oxide; too low and the process slows, producing a thin, porous layer with poor corrosion resistance.
Racking and Contact Issues
Parts must be properly supported in the bath to ensure good electrical contact and consistent current distribution. Poor racking leads to contact marks, uneven coating thickness, and rejected parts.
Sealing Failures
Inadequate sealing is one of the leading causes of corrosion failure in the field. It is also one of the hardest defects to detect visually parts may look perfectly fine but fail a salt spray test within hours.
How AI Anodising Technology Is Modernising the Anodising Process
The quality challenges described above have one thing in common: they are all caused by process variables that drift over time in ways that are difficult for a human operator to monitor continuously. This is precisely where AI anodising technology delivers transformational value.
Real-Time Process Monitoring
AI-powered platforms like AluMind continuously analyse data from sensors embedded across the production line — bath temperature, acid concentration, current density, rectifier performance, and more. Deviations are detected in milliseconds, not after the batch is complete.
Predictive Quality Control
Machine learning models trained on production data can predict with high confidence whether an in-progress batch will meet specification-before it completes. Operators receive actionable alerts and suggested parameter adjustments, preventing defective output rather than simply detecting it.
Energy Optimisation
AI systems can dynamically adjust current profiles and bath timing to achieve the required oxide thickness using the minimum energy input. This reduces both operating costs and carbon emissions — a growing priority as environmental regulations tighten.
Conclusion: Mastering the Anodising Process Starts Here
The anodising process is one of the most reliable and versatile forms of aluminium surface treatment available but achieving consistent quality at scale demands close attention to process variables that are difficult to manage manually. From understanding the difference between Type I, II, and III anodising to recognising where quality breaks down, the fundamentals covered in this guide give you a solid foundation.
The next frontier is using that knowledge in combination with AI-driven monitoring and predictive control. Manufacturers who make this transition are not just improving quality they are reducing costs, cutting waste, and building a competitive edge that is hard to replicate.
AluMind is purpose-built for this challenge. Our AI-powered platform integrates directly with your existing anodising line, giving you real-time visibility and intelligent control without disrupting production.
Ready to see what AI can do for your anodising operation? Talk to the AluMind team today and discover how intelligent process control can raise your quality standards, reduce energy costs, and protect your margins.
Book a Free DemoFrequently Asked Questions
Painting applies a coating on top of the metal surface that can peel or chip over time. Anodising is an electrochemical process that converts the aluminium surface itself into a protective oxide layer — it cannot peel because it is part of the metal. Anodised finishes are generally more durable, more dimensionally stable, and more recyclable than paint.
Thickness depends on the type of anodising. Type I (chromic acid) produces 0.5–2.5 microns; Type II (sulphuric acid) typically produces 5–25 microns; and Type III (hardcoat) can reach 25–150 microns. For context, a human hair is approximately 70 microns in diameter.
Anodising is most commonly applied to aluminium, which naturally forms a thin oxide layer on its own. The anodising process thickens and hardens this layer significantly. Other metals such as titanium, magnesium, and zinc can also be anodised, but aluminium anodising is by far the most commercially widespread application.
Key tests include coating thickness measurement (using eddy current or cross-section microscopy), salt spray testing for corrosion resistance, seal quality assessment using the acid dissolution test, and abrasion resistance testing. The relevant standards for aluminium surface treatment include BS EN 12373 (European) and MIL-A-8625 (US military specification).
Yes — and the evidence is compelling. AI systems monitor real-time process data continuously across every variable, which no human operator can do at scale. Platforms like AluMind use machine learning to predict defects before they occur and recommend parameter corrections automatically, achieving defect prediction accuracy of up to 98%. This translates directly into fewer rejected batches, lower rework costs, and more consistent output.